This is the second post in our Surfactant 101 series. In this post, we’ll be discussing the basics of nonionic surfactants and there use in a wide range of applications.
No Electrical Charge
The nonionic category of surfactants are chemical structures that contain active molecules with no electrical charge. Unlike molecules with surface properties due to a positive (cationic) or negative (anionic) charge, nonionic surfactants do not react with other ions. As a result, they do not form insoluble salts, can be used in strong acidic solutions and tend to have low toxicity profiles.
In a nutshell, nonionic surfactants are utilized in formulations for their resistance to water hardness deactivation, excellent grease removing capabilities, preferable environmental and safety profiles, and compatibility with other surfactants.
Detergents, Cleaners, Industrial Applications
Nonionics are found in everyday products used in households, including personal care items, cosmetic formulations and laundry and dish detergents, as well as commercial scale industrial applications in environmental protection, paper processing and thin films.
Depending on the application, a nonionic surfactant will often serve a specific purpose in the formulation. For instance, they are used as defoaming aids in automatic dish washing, oil recovery agents in clean up operations, and wetting and dispersing agents for soil and turf treatment.
Alcohol Ethoxylates
A common type of nonionic surfactant used in a wide variety of applications are ethoxylated alcohols. Alcohol ethoxylates are known for their positive characteristics in HI&I and industrial applications, such as low toxicity, high biodegradability, varying ethoxylation ranges and excellent cleaning performance.
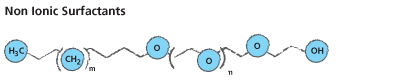
Alcohol ethoxylates are also seen as ideal substitutes for alkylphenol ethoxylates (nonylphenol ethoxylates) due to their lower toxicity risks and higher biodegradability rates. Alcohol ethoxylates may be derived from synthetic or natural sources (i.e. fatty alchohol ethoxylates). A typical fatty alcohol ethoxylate structure will look like the following:
For more information about nonionic surfactants in general, or how alcohol ethoxylates can enhance the performance of a particular formulation, feel free to contact us.
. . . and stay tuned for our next post in the Surfactant 101 series on Amphoterics